The Ozone Layer
Ozone (O3) is made naturally in the atmosphere when three oxygen atoms join together to form a colorless gas.
The earth is wrapped in layers of air called the atmosphere. Stratospheric ozone is found in the stratosphere, the second layer of the earth's atmosphere, 10 to 30 miles above the surface. Life couldn't exist without this protective ozone, which is also called the “ozone layer.”
The sun gives off light, heat, and other types of radiation. Too much UV (ultraviolet) radiation can cause skin cancer, cataracts, and harm plants and animals. Ozone high in the atmosphere absorbs, or takes in, some of the sun's harmful UV rays before they reach the ground. Just as sunscreen helps protect your skin from getting burned, ozone in the stratosphere works like earth's sunscreen.
The Ozone Hole
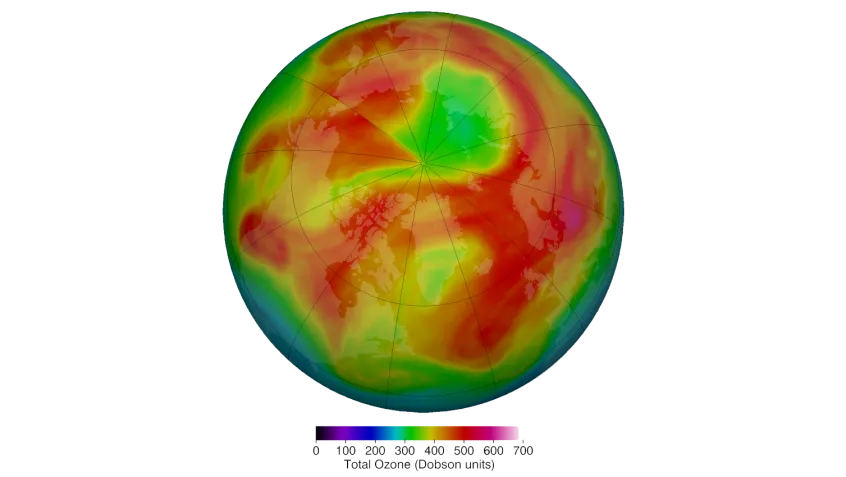
Although we say there is a “hole in the ozone layer” or “ozone hole,” there's no actual hole. Instead, the protective layer contains less ozone than it used to. This thinning is found all over the earth, but the biggest losses are over the North and South Poles. That's because ozone destruction is worse when it's very cold. You can see current levels of ozone over the South Pole at ozonewatch.gsfc.nasa.gov.
The trouble with ozone destruction starts when certain chemicals used in air conditioners, fire extinguishers, insulating foams, and solvents are let out during use. These chemicals eventually reach the upper atmosphere and are broken down by the sun's radiation, releasing chlorine and bromine atoms. These atoms take away one of the oxygen atoms from ozone and use them to make other substances.
Chlorine and bromine atoms are catalysts, meaning they can speed up a chemical reaction without changing, and can repeat the destructive cycle again with another ozone molecule. So one chlorine or bromine atom can destroy thousands and thousands of ozone molecules, causing ozone to disappear much faster than nature can replace it.
Is Anyone Doing Anything about the Ozone Hole (That's Not a Hole)?
The Montreal Protocol is an international treaty that protects the ozone layer by phasing out the manufacture and use of ozone-depleting chemicals. It was enacted in 1989 and all of the countries in the world have signed it. Many ozone-depleting chemicals are now illegal to use, or are only used in small quantities. Because of the Montreal Protocol, levels of most ozone-depleting chemicals in the atmosphere have slowly gone down. Based on current trends, scientists today expect the hole to return to 1980 levels by 2070.
Today, any products in the US containing chlorofluorocarbons (CFCs) and other ozone-depleting chemicals must have warning labels. The US also prohibits the release of refrigerants used in car and home air conditioners into the air because they still use ozone-depleting chemicals.
Learn more from NASA in the video below: For the first time, scientists have shown through direct satellite observations of the ozone hole that levels of ozone-destroying chlorine are declining, resulting in less ozone depletion.
Why Can't We Just Make More Ozone?
Ozone molecules are constantly being made and destroyed by the sun's ultraviolet light in natural processes. Normally, the amount made and the amount destroyed is about the same, so nothing changes. Think of the amount of ozone as the water level in a bathtub with the faucet running and the drain open. If you turn on the water just right, you can make the amount of water leaving the bathtub equal to the amount coming in, so that the water level never changes. But right now, the drain has gotten faster, and the amount of ozone destroyed is more than the ozone being made.
A big reason we can't make more ozone to send into the upper atmosphere is because it would take a LOT of energy. In the atmosphere, this huge amount of energy comes from the sun. We also don't have a way to transport the ozone to the right places in the atmosphere.
Since we can't make more ozone, the solution is to slow the flow down the drain back to its normal rate. And the only way to do that is to stop using ozone-depleting chemicals.
Learn More
Visit NASA's Ozone page to check out the latest science and stories about ozone and the ozone hole. To see the latest status of the ozone hole over Antarctica, visit NASA's Ozone Watch.
This information is adapted from NEEF's SunWise program. Learn more about SunWise.


